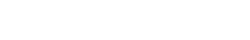NativeSkin® maintains normal human skin integrity and metabolism during 10 days in culture
USING NATIVESKIN EX VIVO SKIN MODEL
Research by E. Raude (Genoskin), E. Pagès (Genoskin), M. Pastore (Genoskin), P.-O. Cuoc (Promega), L. Malaquin (LAAS-CNRS), P. Descargues (Genoskin)
Human epidermis is not vascularized and oxygen only reaches epidermal cells by diffusing through the stratum corneum or from the dermal papillae which contains a dense capillary network. It is known that the main bioenergetic pathway of human epidermis is anaerobic glycolysis (Ronquist et al., 2003). Therefore, glucose consumption and high production of lactate indicate an active metabolism in skin. This pathway has not been well investigated in human skin biopsies cultured ex vivo. Genoskin has developed NativeSkin®, a standardized ex vivo human skin model containing both epidermis and dermis, produced with discarded skin samples. The skin model is embedded in a proprietary nourishing gel-like matrix with epidermal surface left in direct contact with the air. NativeSkin® represents a highly predictive and cost-effective last-line screening tool in laboratory conditions prior to in vivo clinical evaluations. We study here the integrity, viability, and metabolism of NativeSkin® models after 10 days of ex vivo culture.
NativeSkin® shows cell proliferation and no apoptosis at day 10
Anti-Ki67 and anti-active Caspase-3 immunostainings were performed on 5µm thickness skin slices to assess cell proliferation and apoptosis. Representative images were presented. Quantifications were performed on 6 representative pictures of the epidermis.
37% of cell proliferation were maintained after 10 days of ex vivo culture. Depending on the donor, levels of proliferation were from 22 to 69% of levels just after surgery. Levels of apoptosis marker were low after 10 days of ex vivo culture, with only 0.15 to 2% of the total epidermis area stained.
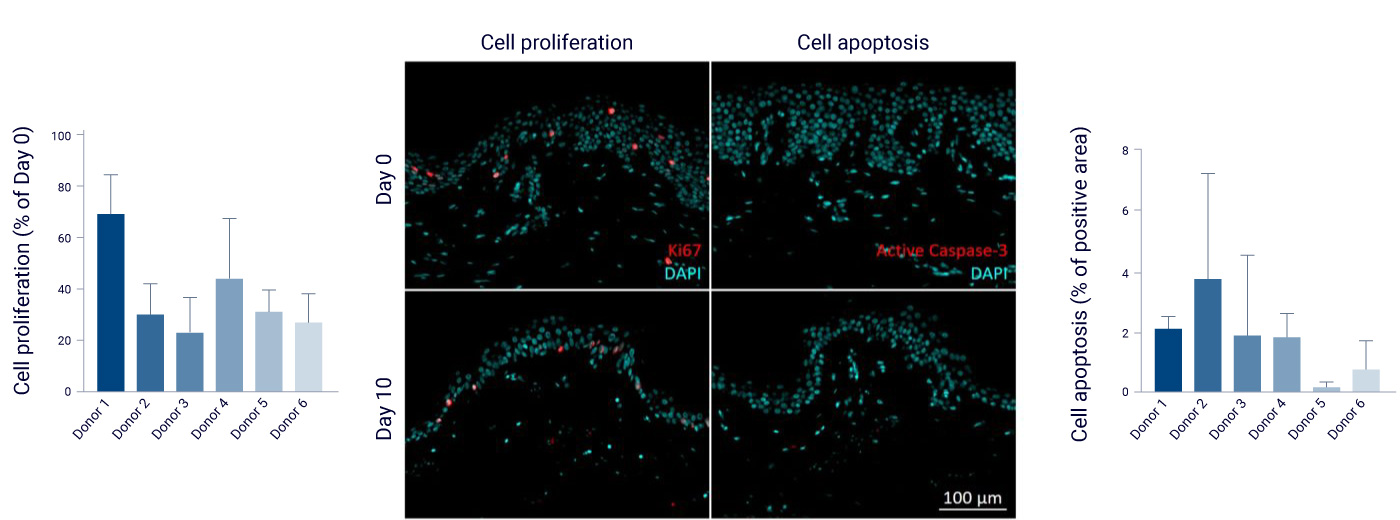
Anti-KI67 and anti-active caspase-3 immunostainings and quantification.
NativeSkin® maintains normal skin integrity and a significant cell viability at day 10
Hematoxylin and Eosin staining was performed on 5µm thick skin slices. A representative image of NativeSkin® model at Day 0 and Day 10 is presented for each donor.
Observations of histological staining revealed that the skin integrity was preserved after 10 days of ex vivo culture.
Normal skin integrity after 10 days of ex vivo culture.
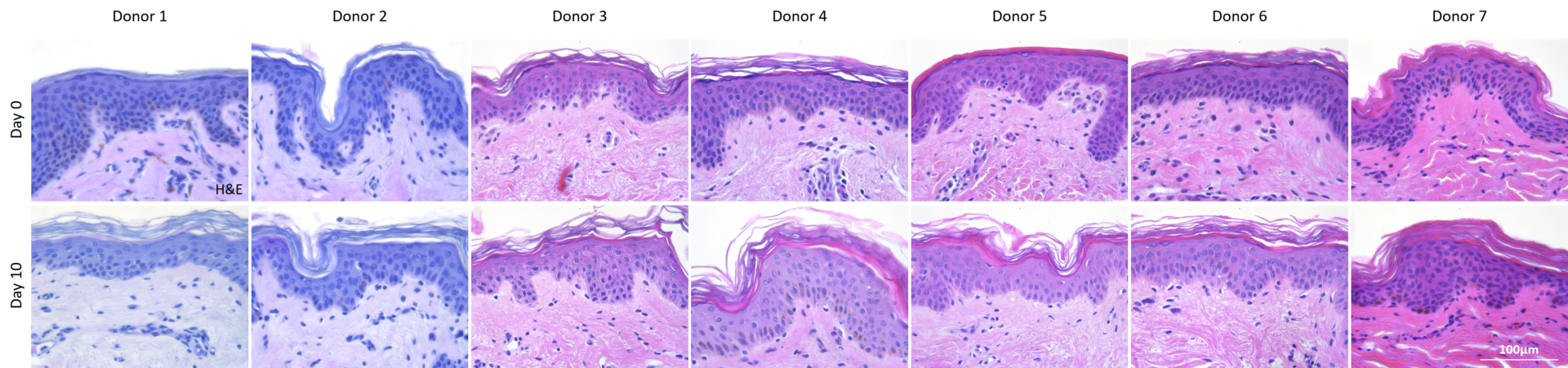
WST-8 assay was performed on small punch biopsies (2 in the center and 4 in the edge), in order to assess cell viability in different area of the model.
WST-8 measurements demonstrated that tissue was viable after 10 days of ex vivo culture, with values from 44 to 77 % of initial viability depending on the donor. Viability in the center or in the edge of the biopsy were not significantly different differences.
Cell viability after 10 days of ex vivo culture.
NativeSkin® consumes glucose and produces lactate at day 10
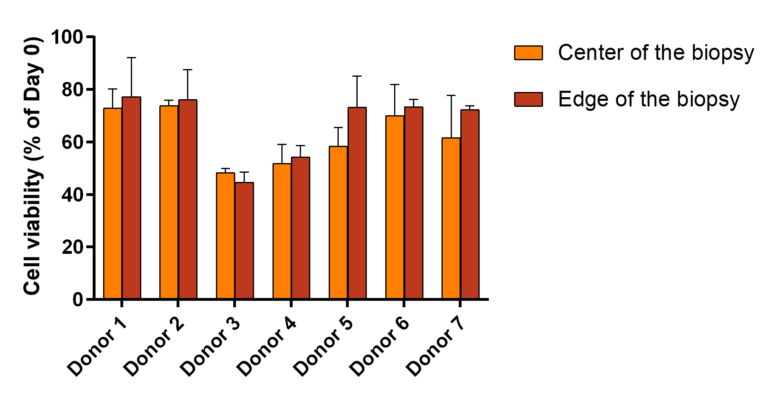
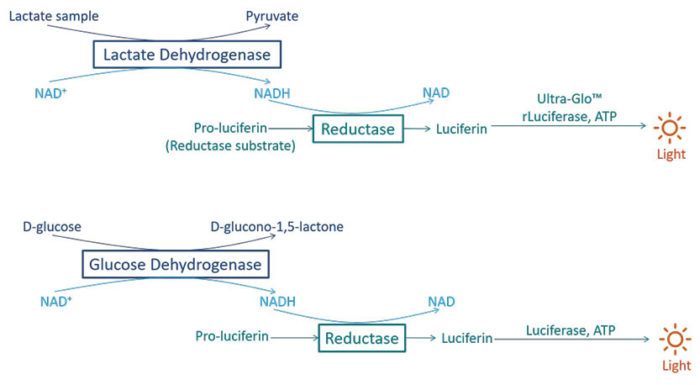
Skin metabolism was analyzed on both culture media and biopsies after 10 days of ex vivo culture, by dosing two essential metabolites: lactate and glucose. Dosing were performed using Promega bioluminescent NADH assays, Lactate-Glo™ Assay and Glucose-Glo™ Assay. We followed protocols recommended by the manufacturer.
Dosing of glucose and lactate. (Illustration based on Promega figure)
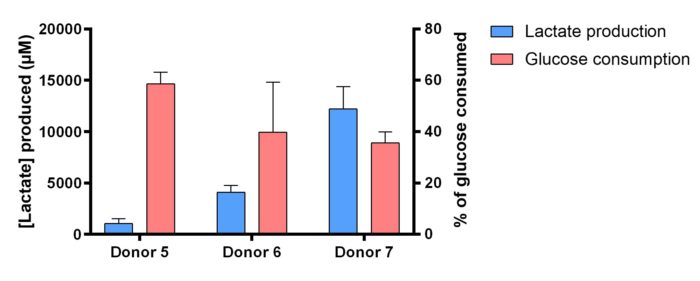
Concentrations of lactate produced and glucose consumed in culture media between Day 9 and Day 10 were dosed. The bars represent the mean ± SD of replicates.
Lactate production and glucose consumption in culture media.
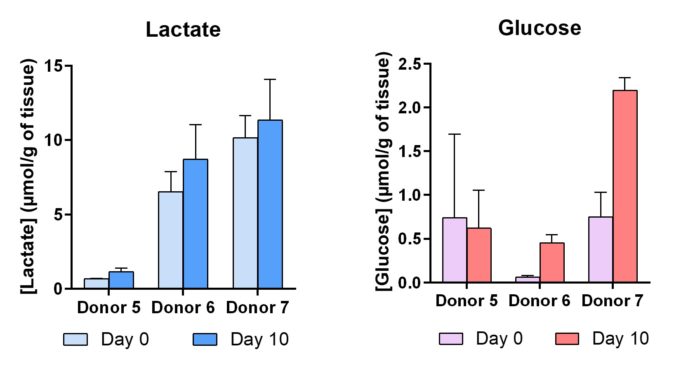
Lactate and glucose concentrations in NativeSkin® cultured 10 days were measured on 1 frozen biopsy for each sample. The bars represent the mean ± SD of replicates.
Lactate production and glucose consumption.
Conclusion
NativeSkin® is an ex vivo human skin model that can normally be maintained in standard culture conditions up to 7 days when shipped to customers. Here, we demonstrate for the first time maintaining the skin model 10 days in ex vivo culture while preserving normal skin integrity, significant cell viability, proliferative cells and low levels of apoptosis.
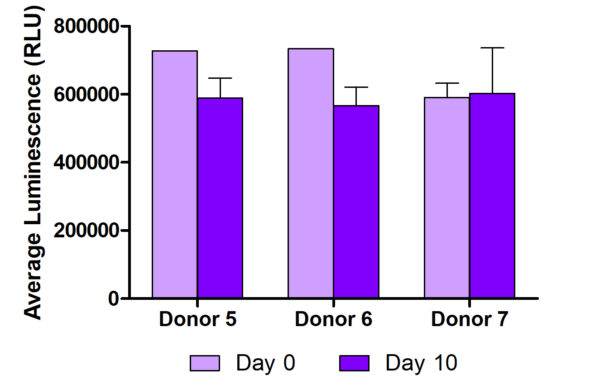
We demonstrate that the skin metabolism of NativeSkin® at 10 days is comparable to fresh skin by measuring glucose consumption and lactate production using Promega’s Glucose-Glo™ and Lactate-Glo™ Assays. We also demonstrate that NativeSkin® maintains normal ROS concentration after 10 days of ex vivo culture, using Promega’s ROS-Glo™ H2O2 Assay. Our next step will be to evaluate the activity of specific human skin cytochromes involved in the metabolization of drugs topically applied on NativeSkin®.